e-ISSN: 2321-6182 p-ISSN: 2347-2332
e-ISSN: 2321-6182 p-ISSN: 2347-2332
Warra AA*1, Umar RA2, Sani I1, Gafar MK3, Nasiru A4, and Ado A5
1 Dept. of Biochemistry, Kebbi State University of Sci. & Tech. P.M.B. 1144, Aliero, Nigeria.
2 Department of Biochemistry, Usmanu Danfodiyo University, PMB 2346, Sokoto, Nigeria.
3 Department of Chemistry, Federal University, Dutsin-Ma, Katsina State, Nigeria.
4 Department of Biochemistry, Bayero University, P.M.B. 3011, Kano, Nigeria.
5 Ministry of Education Science and Technology, Dutse, Jigawa State, Nigeria.
Received: 25/07/2013 Revised: 10/08/2013 Accepted: 22/09/2013
Visit for more related articles at Research & Reviews: Journal of Pharmacognosy and Phytochemistry
This work was carried out to determine the potential applications of gingerbread plum seed oil by investigating its phytoconstituents and physicochemical characteristics. The underutilize oil seed from gingerbread plum tree growing in abundance in Niger Republic was exploited. Phytochemical analysis of the seed oil extracted using soxhlet apparatus with n-hexane revealed the presence of saponins, alkaloids, flavonoids, steroids, terpenoids, and tannins and absence of Cyanogenic glycosides.The physicochemical analysis of gingerbread plum (Parinari macrophylla) seed oil was carried out and the following results were obtained; Acid value 12.97± 0.01 mg KOH/g, Saponification value 153.30 ± 0.10 , Iodine value 32.07 ±0.01 I2/100g, Free fatty acid 15.10±0.10 ( % Oleic), Peroxide value (meq H2O2) 45.48 ± 0.02. The % yield was 49.3, Color was golden yellow, odour was agreeable, and the oil was liquid at room temperature.Justification of the use of the seed oil for food, medicinal and cosmetic was expatiated.
gingerbread plum seeds, oil extraction, phytochemical, physicochemical, medicinal.
Parinari macrophylla (Sabine) Chrysobalanaceae, has been use extensively in the Northern part of Nigeria in Ethnomedicine. It is used to treat numerous diseases which include: Asthma, Skin infections, treatment of wounds, pulmonary troubles, dysentery, inflammations and it is also used for the treatment of eye and ear infections [1]. It is known in Hausa Language as ‘Gawasa’ [2,3] the plant is semi-cultivated [4]. A purely West African species (formerly Parinari macrophylla Sabine;now Neocarya macrophylla (Sabine) Prance). Its mealy fruits are especially loved in Sierra Leone, but are esteemed from Senegal to northern Nigeria, where they can be seen in the local markets. The flesh is soft and yellowish when fresh, with a peculiar flavour sometimes likened to avocado. It is esteemed for many additional products. The rind from fresh fruits is used to impart a pleasant scent to ointments. The living tree provides villagers with dye, glue, fodder, firewood, soap, structural materials, and even termite repellents (in the Gambia). And the leaves are used medicinally for such things as toothache and mouthwash [5]. Extraction and saponification of Gingerbread plum (Parinari macrophylla) Seed oil was reported [6]. The aim of this research was to determine the potential applications of gingerbread plum seed oil in foods and cosmetics its phytoconstituents and physicochemical characteristics.
Seed material
Parinari macrophylla (Rosaceae) seeds obtained from Gingerbread plum tree were collected in the Month of October from Junju town, Niger Republic. The other parts of the plant collected were the leaves, the fruits and the flowers for the purpose of identification. The plant was identified and authenticated by a Botanist at the Biological Sciences Department, Bayero University, Kano (BUK) Nigeria. Confirmation of taxonomic identity of the plant was achieved by comparison with voucher specimen (voucher No. 175) kept at the Herbarium of the Department of Biological Sciences and use of documented literature [7]. The seeds were selected and damaged ones were discarded. The seeds were cleaned, de-shelled and well dried and ground using laboratory plastic pestle and Mortar prior to extraction.
Oil extraction
The routine extraction of 35g of grounded seeds of Parinari macrophylla (Rosaceae) was conducted in a soxhlet extractor using n-hexane (boiling between 40–60°C) for six hours. The oil was obtained after the solvent was removed under reduced temperature and pressure and refluxing at 70 °C to remove excess solvent used in the oil. Extracted seed oil was stored in freezer at–2 °C for subsequent physicochemical analysis. The extraction was carried out in the Biochemistry Laboratory, Department of Biochemistry, Kebbi State University of Science & Technology, Aliero, Nigeria.
Oil Yield
The oil which was recovered by complete distilling of most of the solvent on a heating mantle was then transferred to measuring cylinder. The measuring cylinder is then placed over water bath for complete evaporation of solvent for about 2-3 hours in accordance with the method reported [8] and weight of the oil was recorded and expressed as oil content(%) as follow
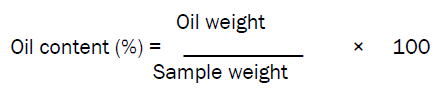
Qualitative Phytochemical Analysis
The method reported in literature [9] was used.
Test for tannins
Few drops of 1% lead acetate were added to 5ml of the oil extract in a test tube. A yellow precipitate was formed which indicated the presence of tannins.
Test for saponins
The oil extract was diluted with 2 ml of distilled water and it was agitated in a test tube for about 15 minutes. The formation of 0.1cm layer of foam showed the presence of saponins.
Test for flavonoids
Few drops of dilute sodium hydroxide were added to 1ml of the oil extract in a test tube. An intense yellow color was formed which turned colorless on addition of few drops of dilute acid indicating the presence of flavonoids.
Test for alkaloids
The oil extract (2ml) was added to 2ml of HCl. To the acidic medium, 1 ml of Dragendroff’s reagent was added. An orange or red precipitate was immediately formed which indicated the presence of alkaloids.
Test for steroids
To 1ml of the oil extract in a test tube, 10 ml of chloroform was added. Equal volume of concentrated sulphuric acid was added by the sides of the test tube. The upper layer turned red, whereas the sulphuric acid layer turned yellow with green fluorescence. This indicated the presence of steroids.
Test for terpenoids
Two ml of the oil extract was mixed in 2 ml of chloroform, and concentrated H2SO4 (1 ml) was carefully added to form a layer. A reddish brown coloration was formed at the interface to show positive results for the presence of terpenoids.
Test for cyanogenic glycosides
Small quantity of the oil extract was put in a test tube. 1.5ml of distilled water and 6 drops of chloroform were added and the mixture stirred with a rod. The test tube was stoppered with a cork containing a strip of picrate-impregnated paper hanging down from the stopper, and incubated at ambient temperature for 2 hours. A color change of the paper, from yellow to brown-red, indicated the release of HCN by the plant. If there was no release of HCN within 2 hours, indicating a negative test, the tube was left at ambient temperature for 24 and 48 hours, so that it could be re-examined. A brown-red coloration within 2 h indicated the presence of cyanogenic glycoside and the respective hydrolytic enzyme, and the plants were considered cyanogenic in the field. A brown-red color appearing within 48 hours indicated that the cyanogenic glycoside spontaneously released HCN without the action of enzyme. No color change after 48 hours indicated that the test was negative for cyanogenic glycoside.
Physicochemical analysis
The acid value, iodine value and saponification value determinations were carried out using the methods reported [10,11,12]. Free fatty acid was determined according to the Official Method Ca 5a-40 [13] while peroxide value was determined using the method specified by International standard organization [14].
The results of qualitative phytochemical analysis on the oil extract of Parinari macrophylla seed oil are shown in Table1. The results obtained showed the presence of Tannins Saponins, Flavonoids, Alkaloids , Steroids and Terpenoids.
The phytochemicals found in the seeds oil of this plant are relatively similar to those in Anacardium occidentale L. seeds oil which have been reported to have contained antimicrobial agents [9].
Flavonoids are slightly present. The flavonoids are a remarkable group of plant metabolites. No other class of secondary product has been credited with so many–or such diverse–key functions in plant growth and development. The flavonoids are evidently extremely useful to plants, and it is not surprising, therefore, that species from all orders of the plant kingdom, from the basal liverworts to the most advanced angiosperms, invest significant amounts of metabolic energy into the production of these compounds [15]. Two main subsets are known that perform different functions, the colourful red and blue flavonoids (anthocyanins and 3- deoxyanthocyanins), the anthocyanin molecules absorb green light and affords unique capabilities, such as the protection of chloroplasts from the damaging effects of strong irradiance, and as a visible cue to some animals. Flavonols and flavones, on the other hand ( i.e. colourless or yellow flavonoids), do not directly affect photosynthesis, but they can act as chemical signals or UV guides to attract or deter insects, and are highly effective UV filters.
Flavonoids are now recognized as possessing an array of bioactivities with several mechanisms relevant to potential reductions in the pathogenesis of chronic diseases (e.g., anti-inflammatory and antioxidant actions as well as alteration of redox-sensitive signal transduction pathways and gene expression) [16].
Tannins were slightly present. Generally Tannins have different functions in that they serve as chelating agents for metals ion, antioxidants in biological systems, and as protein precipitating agents
Saponins were largely present in the seed oil sample. Because of their surfactant nature, they are used industrially in mining and ore separation, in preparation of emulsions for photographic films, and, extensively, in cosmetics, such as cleansing formulae. In addition to their emollient effects, the antifungal and antibacterial properties of saponins are important in cosmetic applications [17].
The presence of terpenoids was found to be largely in Parinari macrophylla seed oil extract justifies why decoction of dried kernel can used for the treatment of diabetes mellitus as reported for Cashew nut oil [9].
Among the secondary metabolites that are produced by plants, alkaloids figure as a very prominent class of defense compounds [18]. Alkaloids were found largely present in Parinari macrophylla seed oil extract which signifies their functionality in repellence, deterrence, toxicity and growth inhibition by herbivores/ predators and in growth inhibition and toxicity by microbes/ viruses and as secondary metabolites for UV- Protection and Nitrogen storage.
The steroids are modified triterpenoids containing the tetracyclic ring system of lanosterol (Figure1), but lacking the three methyl groups at C-4 and C-14. Cholesterol (Figure 2) typifies the fundamental structure, but further modifications, especially to the side-chain, help to create a wide range of biologically important natural products, e.g. sterols, steroidal saponins, cardioactive glycosides, bile acids, corticosteroids, and mammalian sex hormones. Because of the profound biological activities encountered, many natural steroids and a considerable number of synthetic and semi-synthetic steroidal compounds are routinely employed in medicine [19].
The steroids were averagely present in Parinari macrophylla seed oil extract which signifies its various medicinal significance.
Figure 1: Tetracyclic ring system of lanosterol [19].
Figure 2: [19].
The physicochemical analysis of the gingerbread plum (Parinari macrophylla) seed oil showed that it has saponification value of 153.30±0.10 KOH/g which is lower than the saponification value of the the following seed oil; Citrus lanatus 189.35mgKOH/g [20], Adansonia digitata linn seed oil 230.01 mg KOH/g [21], Elaeis guineensis seed oil 246.60 mg KOH/g [22], higher than the saponification value of the following seed oils; Polyalthia longifolia seed oil 120.00 mg KOH/g [23], Cashew karnel oil 137 mg KOH/g [24], Lagenaria siceraria seed oil [25]. High saponification value indicates or justifies the usage of oil for soap making [26].
Iodine value was 32.07 ± 0.01g I2/100g which is lower than the iodine value of the following oils; [27], Hypertis spicigera seed oil 81.22I2/100g [28], higher than the iodine value of Landolphia owariensis seed oil 15.10I2/100g [29], The iodine value that was obtained is below 100. Oils with iodine value below 100 are called Non - drying oils and they are good or useful in soap production [30].
The acid value obtained was 12.97 ± 0.01 mg KOH/g which is lower than the following value obtained from other seed oils; Monodora myristica gaertin seed oil 14.31mgKOH/g [21], rubber oil 15.03 mg KOH/g [31].The acid value is higher than the following seed oils; Hyptis spicigera seed oil 2.5mgKOH/g [28] Citrus lanatus seed oil 5.25mg KOH/g [20] and 10.493 mg KOH/g reported [27] for olive oil is useful in soap making.
The free fatty acid obtained from the analysis 15. 10 ± 0.10 which is lower than the value of callophyllum inophyllum linn seed oil 160.3% [21], higher than the values of following seed oils; polyalthia longifera seed oil 7.73% [23], monodora myristica gaeth dunat 7.20% [21], rubber seed oil 7.55% [31]. Free fatty acid can stimulate oxidative deterioration of oils by enzymatic and/or chemical oxidation to form off flavor component [21].
The peroxide value obtained which has a value of 45.48± 0.02 mgmeq/kg is lower than polyalthia longifera seed oil 730.00mgmeq/kg [23], butyrospermum parkii oil 77.5mgmeq/mg [32]. But higher than; dacroyodes edulis (G.Don) H.J lam seed oil 20.00mg meq/kg [20], stercula setegera seed oil 35.0 mg meq/kg [32]. Higher peroxide value is very likely in products that do not contain preservatives, either natural or synthetic [33]. This signifies that the gingerbread plum seed oil maintains its natural form at the time the analysis was carried out.
Conclusively gingerbread plum seed oil appears to be potential seed oil for industrial applications, it can be recommended for use in food, medicinal and cosmetic preparations.
The authors wish to acknowledge the contribution of Wahida of the Department of Biochemistry, Kebbi State University of Science and Technology, Aliero, Nigeria.