ISSN: 2322-0066
ISSN: 2322-0066
Lan Zhang1,2,3, Ning Xu1, Jianming Man1, Luo Rui1*
1 Department of Life Sciences, Guizhou University, Guizhou Province, China
2 Key laboratory of Plant Resource Conservation and Germplasm Innovation in Mountainous Region (Ministry of Education), Guizhou University, Guiyang 550025, China
3 Collaborative Innovation Center for Mountain Ecology and Agro-Bioengineering (CICMEAB), Guizhou University, Guizhou Province, China
Received: 23-May-2023, Manuscript No. JOB-23-104407; Editor assigned: 25-May-2023, PreQC No. JOB-23-104407 (PQ); Reviewed: 12-Jun-2023, QC No. JOB-23-104407; Revised: 15-Jun-2023, Manuscript No. JOB-23-104407 (R); Published: 22-Jun-2023, DOI: 10.4172/2322-0066.11.2.010
Citation: Zhang L, et al. A Non-Tissue Culture-Dependent Approach for Genetic Transformation of Marchantia polymorpha L. RRJ Biol. 2023;11:010
Copyright: © 2023 Zhang L, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Research & Reviews: Research Journal of Biology
Marchantia polymorpha L. is a prime example of an early terrestrial plant. It acts as a desirable model species for biological research as a result of its short growth cycle, predominant haploid gametophyte, simultaneous sexual and asexual propagation, etc. In this study, a non-tissue culture-dependent approach to the genetic transformation of M. polymorpha L. was developed. This approach comprises immersion of explants (thalli or mature gemmae obtained in the field conveniently, with no pre-culture) in the solution of Agrobacterium, inoculation on soil simply, and screening by spraying selection solution directly on them. As a result, over-expression of MpCCAAT-NFYB5 (Mapoly0053s0055.1) and corresponding phenotypic changes were observed. Furthermore, it was demonstrated that non-chimera transformants could be obtained rapidly through four cycles of gemmae reproduction. This method would facilitate further molecular biological research on this specific plant
Marchantia polymorpha L; Agrobacterium tumefaciens; Non-tissue culture dependent transformation; Non-chimera transformant; MpCCAAT-NFYB5
Marchantia polymorpha L. (Marchantiaceae), which is a member of the earliest land plant species, was recognized long ago; it could be used as a model in many studies related to plant science to explore the mechanism of sexual reproduction, reveal the developmental characteristics at cellular level, etc [1-5]. M. polymorpha L. possesses several advantages for experimental operation in comparison to other model plants, such as easy culture in field (with soil) and laboratory (in vitro culture), short life cycle, small size of the haploid body, propagule formation through sexual and asexual pathways, etc. Furthermore, the publications of whole genome sequences for M. polymorpha L. and M. inflexa and the establishment of two open databases, such as the MarpolBase for M. polymorpha Tak-1 strain and the MarpoDB for M. polymorpha Cam-1 strain have encouraged further research on this organism [6-8]. The production of transgenic organisms might be an option to address the requirements of humankind in agriculture and biological engineering or to determine gene function in biological research. These organisms (genetically modified or transgenic) could be obtained through various strategies or methods [9,10]. Genetic transformations by particle bombardment (microprojectile bombardment or biolistic bombardment) and Agrobacterium-mediated methods have been reported in M. polymorpha L. with stable or transient expressions.
Various plant materials have been employed as receptors in different genetic transformations of M. polymorpha L. by particle bombardment method, such as suspension-cultured cells, whole thalli that originated from gemmae culture after two weeks, or spore culture after seven days or fragment of mature thalli from a gemmae culture with 2.5 to 3 weeks [11-15].
Protocols (with minor modifications) from Takenaka et al. had been employed to study fatty acid metabolism and generate mutants in M. polymorpha L. The Agrobacterium-mediated approach, which was applied first in M. polymorpha L. by Nasu et al. gained increased attention as a result of its convenient operations and minimal requirement of equipment [16-19]. All protocols based on Agrobacterium should be divided into two categories, such as those that involve infection of plant materials by co-cultured Agrobacterium in liquid system or on the surface of the solid medium in a petri dish [20-28]. In the genetic transformation of M. polymorpha L., various plant materials, which were suspension-cultured cells, whole thalli from spores (sporelings) or gemmae (gemmalings), or pieces from immature or mature thalli, could also function as receptors. In various studies, different protocols of Agrobacterium-mediated genetic transformation were applied to reveal the characteristics of the auxin-transcriptional response system and establish genome editing methods in M. polymorpha L [29-31].
More efficiently, transformation protocols independent of plant tissue culture have been established, such as floral-dip transformation of Arabidopsis thaliana, ovary-injection transferring of Paphiopedilum Maudiae (a cross between Paphiopedilum calosum x Paphiopedilum lawrenceanum), in planta transformation of Notocactus scopa cv. Soonjung, Kalanchoe pinnata, etc [32-35]. Recently, Cao et al. developed the Cut-Dip-Budding (CDB) genetic delivery system and transformed six kinds of plant species without involving sterile tissue cultures on Petri dishes. However, the protocols for the genetic transformation of M. polymorpha L. (based on particle bombardment or Agrobacterium mediation) still involve in vitro culture. Consequently, the pollution issues associated with co-culture with Agrobacterium frequently interfere with progress. To avoid the troublesome content (i.e., tissue culture sterilely on a petri dish) and make the genetic transformation in M. polymorpha L. simpler and more efficient, we conducted a study involving the delivery of genetic material directly into thalli or gemmae collected from soil culture individuals. Our results demonstrate that effective genetic modifications could be performed without tissue culture on petri dishes. These findings could be beneficial for future molecular research concerning M. polymorpha L.
Plant material and growth conditions
M. polymorpha L. was obtained from its natural environment (Huaxi, Guiyang, China. 106.6592°E, 26.4496°N; Alt. 1050 m) and cultivated in a garden with moderate humidity levels. Thalli that were cultured for three or four weeks and mature gemmae that could disperse from gemmae cups spontaneously were excised and collected, respectively, for genetic transformation. The soil used to grow the liverwort was garden soil obtained from its wild habitat. Soil sterilization and closed-culture with containers plastic box with lid, Figure 1 were adopted to avoid contamination from algae, microorganisms, or dispersed gemmae of other liverworts and to maintain soil humidity by control of water evaporation. Plants in soil culture were grown in the shade (without direct sunlight, natural rainfall, and natural temperatures) (Figure 1).
Sensitivity test of M. polymorpha L. to kanamycin
Tests of sensitivity to kanamycin were performed on closed soil culture or solution culture (in petri dishes). For closed soil culture, individuals developed from gemmae were treated with selection buffer (containing cefotaxime and various concentrations of kanamycin) following two weeks in the culture. The selection buffer contained cefotaxime (1.0 mg·mL-1) as well as varying concentrations of kanamycin (1.0, 2.5, 5.0, 7.5, 9.0, 10.0, or 11.5 mg·mL-1). Screen solution was sprayed on soil and plant surfaces every two days. For solution culture, gemmae were cultured in liquid 1/2 B5 medium supplement without sucrose and with antibiotics (1.0 mg.mL-1 cefotaxime and 0.25, 0.50, 0.75, or 1.00 mg·mL-1 kanamycin). The number of survival individuals for all treatments was counted following two weeks of treatments with various selection buffers.
Plasmid and A. tumefaciens strains
The plasmid pBI121, which possesses kanamycin-resistant gene (selection marker) and GUS gene (expression label), was employed as a binary vector for carrying MpCCAAT-NFYB5 (Mp6 g07410.1), which is a transcriptional factor from nuclear factor Y family, under the control of a cauliflower mosaic virus 35S promoter. Competent state cells of A. tumefaciens strain GV3101 were prepared [36]. The transformation of pBI121-MpCCAAT-NFYB5 into bacterial cells was performed through the liquid nitrogen freeze-thaw procedure [37]. A single colony of bacteria was selected for liquid culture in Yeast Extract Peptone (YEP) liquid medium (5 mL system), and the transformation was thereafter verified by vector through PCR. The verified positive bacterium (1 mL solution) was subsequently transferred into 50 mL YEP liquid medium in the flask, where it was suspended for 6~8 h at 28ºC with 250 rpm to reach an OD600=0.5~0.6. The bacteria deposit was resuspended in the transformation buffer at OD600=0.5 following centrifugation (6000 rpm, 10 min, RT).
Agrobacterium-mediated direct transformation
Cut pieces of thalli (three or four weeks culture from mature gemmae on soil) or mature gemmae were used as explants for either direct immersion in transformation buffer containing bacteria or through negative-pressure infiltration treatment with injector following manual immersion immediately (Figure 1). Immersed plant materials were transferred to filter paper to absorb liquid on the surface. The treated plant materials were transferred and inoculated on the sterilized soil in cups. Following three days of dark culture and a week (for thalli pieces) or two weeks (for gemmae) of light culture in the garden, growing individuals were screened by two-week spraying of selection buffer containing cefotaxime (1.0 mg·mL-1) and kanamycin (11.5 mg·mL-1) once every two days. Plants that survived were transplanted to fresh soil to grow for 4~6 weeks continuously. Because we were concerned that the insertion of exogenous gene fragments on pBI121 vector would affect the conversion rate, we also carried out the transformation experiment of pBI121 vector to earth. The experimental data showed that there was no significant difference between the conversion rate of pBI121 vector and MpCCAAT-NFYB 5 vector (Table 2).
Identification of transgenic plant by PCR and GUS staining
Surviving plants grown sufficiently large were used for total DNA extraction by Cetyl Trimethyl Ammonium Bromide (CTAB) method. PCR amplification of the GUS gene was performed in 25 µL reaction system using Es Taq MasterMix (Cat# CW0690M, Biopike co.) supplemented with a template (0.25 µg), sense primer (0.4 µM, 5′-TCTGCGACGCTCACACCGAT-3′), and antisense primer (0.4 µM, 5′-GCCAACGCGCAATATGCCTT-3′). The following amplification conditions were used: 94ºC for 4'; 34 cycles of 94ºC for 30", 59ºC for 30", and 72ºC for 2'; final extension at 72ºC for 10'. PCR products were detected using electrophoresis with agarose gel (1%). Positive individuals in PCR identification were selected for Glucuronidase (GUS) staining. Transformed individuals (T0 generation) that were positive in PCR identification and GUS staining were designated specific material numbers and cultured to characterize the phenotype and produce fresh gemmae for further tests.
Continuous culture to produce non-chimera individuals
Transformed individuals of the T0 generation were generally chimeras. Non-chimera individuals were produced using a culture of M. polymorpha L. that was continuously based on the asexual life cycle (from gemmae to thalli repeatedly). Thalli (Tn+1), which were developed from gemmae that produced thalli (Tn) and grew under selection conditions (as described above), were supplied to the extract of total DNA to determine the status of transformation. For one thallus/individual (Tn), the determination of its transformed status was based on next-generation gemmae/thalli (Tn+1) from four gemma cups (at least twelve gemmae for each cup). In the absence of a chimera, the offspring of a single thallus/individual would be viable under selective conditions, exhibit positive PCR identification, and show whole staining instead of local staining in GUS detection.
Quantification of MpCCAAT-NFYB5
Thalli of individuals that were grown from gemmae on soil for approximately four weeks were subjected to total RNA extraction by MolPure® Plant RNA kit (Cat# 9109, TakaRa). Subsequently, cDNA synthesized by Hifair® 1st Strand cDNA Synthesis Kit (gDNA digester plus) and reagent kit from YEASEN (Cat# 11139ES60) were used to quantify expression of MpCCAAT-NFYB5. qRT-PCR was performed on Bio-Rad CFX96 instrument with ChamQ Universal SYBR qPCR Master Mix reagent (Cat# Q711-02, Vazyme) in accordance with manufacturer's instructions in 20 µL reaction system. MpAPT (adenine phosphoribosyl transferase, Mp3 g25140) was selected as an internal reference for the quantification of MpCCAAT-NFYB5. Primers for qRT-PCR were listed as follows: sense primer (0.2 µM, 5′- CGAAAGCCCAAGAAGCTACC-3') and antisense primer (0.2 µM, 5′- GTACCCCCGGTTGCAATAAG-3′) for MpAPT (product length: 150 bp); sense primer (0.2 µM, 5′- ATTCTCCCAAAGCAGGTGGG-3′) and antisense primer (0.2 µM, 5′-GTACGGAGGATGCAGATACACC-3′) for MpCCAAT-NFYB5 (product length: 150 bp). In each analysis, three biological repeats and three technical replicates were used. Amplification conditions were listed as follows: 95ºC for 30'; 40 cycles of 95ºC for 5', and 60ºC for 30'.
Culture under selection conditions could facilitate the identification of positive individuals in genetic transformation. To assess the efficacy of selection conditions independent of tissue culture, kanamycin sensitivity tests for M. polymorpha L. were performed on soil cultures or solution cultures. The results demonstrated that gemmae (i.e., nascent or developing thalli) in solution culture were more sensitive to kanamycin in comparison to individuals (i.e., rapid growth thalli) in soil culture: for solution culture, all gemmae died with 1.0 mg·mL-1 kanamycin, while for soil culture, all thalli expressed normal growth with 1.0 mg·mL-1 kanamycin but died with 11.5 mg·mL-1 kanamycin (Table 1 and Figures 2A-2O).
| Content of kanamycin (mg-ml-1) | 0 | 0.25 | 0.5 | 0.75 | 1 | 2.5 | 5 | 7.5 | 9 | 10 | 11.5 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Death rate (%) | Soil culture | 0 | – | – | – | 0 | 0 | 0.2 | 2.8 | 67.4 | 94.9 | 100 |
| Solution culture | 0 | 24.1 | 69.1 | 87.9 | 100 | – | – | – | – | – | – | |
Note: – represents no test for soil culture or solution culture; the numbers involved in the test for soil culture and solution culture were 175 and 106, respectively. Death rate (%)=(No. of dead/No. of inoculated) × 100.
Table 1. Survival of M. polymorpha L. under different selection conditions.
Figure 2: Growth of thalli in soil culture or gemmae in solution culture under various selection conditions. (A-I) Soil culture for two weeks; (A-H) Growth with 0, 1.0, 2.5, 5.0, 7.5, 9.0, 10.0 or 11.5 mg·mL-1 kanamycin treatments, respectively; (I) Death thallus (rapid growth thallus); (J-O) Solution culture for two weeks; (K-N) Growth with 0, 0.25, 0.5, 0.75, or 1.0 mg·mL-1 kanamycin treatments, respectively; (J) Death gemmae (nascent thallus).
Explants (cuttings of thalli or mature gemmae) were transformed either by direct immersion or through infiltration with negative pressure, followed by an immersion in the injector. The plant materials that had been treated with Agrobacterium were cultured on soil for two weeks and subsequently selected for the next two weeks by spraying selection buffer (11.5 mg·mL-1 kanamycin) once every two days. Effective transformation of M. polymorpha L. could be achieved through these simple protocols.
There were no significant differences among the differential explants regarding survival ratio after selection, regardless of the method or duration of treatment employed. However, for all kinds of explants, a higher ratio of transformant was obtained by negative-pressure infiltration than by immersion alone; and increased time of treatment resulted in a higher ratio of transformant by each of these two kinds of treatment (Table 2). Either the pBI121 vector or the pBI121-MpCCAAT-NFYB 5 vector, Effective transformation of M. polymorpha L. could be achieved through these simple protocols (Table 2), Phenotypic changes occurred in positive transgenic plants with pBI121-MpCCAAT-NFYB 5 vector selected for transformation (Figure 3A).
| Treatments | Times of direct immersion | Times of infiltration with injector | |||||
|---|---|---|---|---|---|---|---|
| Explants | 7 min | 10 min | 15 min | 7 min | 10 min | 15 min | |
| pBI121-MpCCAAT -NFYB5 | T- (3 w) | 66/0/0 | 84/26.2/31.8 | 81/19.8/37.5 | 120/33.3/25.0 | 120/31.7/31.2 | 120/26.7/53.1 |
| T- (4 w) | 72/40.3/6.9 | 72/63.8/15.2 | 72/37.5/22.2 | 120/20.8/24.0 | 120/49.2/32.2 | 120/60.8/58.9 | |
| Gemmae | 240/11.3/11.1 | 240/23.7/15.8 | 240/23.7/19.3 | 394/22.1/24.1 | 411/25.1/30.1 | 407/29.9/41.8 | |
| pBI121 vector | T- (3 w) | 60/1/0 | 63/28.6/33.3 | 66/21.2/35.7 | 127/21.6/28.9 | 123/28.5/34.3 | 120/27.5/57.6 |
| T- (4 w) | 64/39.1/16 | 64/59.4/18.4/ | 64/34.4/27.3 | 120/18.3/27.3/ | 120/41.7/28.0 | 120/56.7/54.4 | |
| Gemmae | 176/11.9/19.0 | 176/24.4/18.6 | 176/22.7/22.5 | 240/19.7/25.5 | 242/21.9/32.1 | 247/25.5/38.1 | |
Note: *Data in the table are numbers of explants/ratio of survival after selection/ratio of transformant identified by PCR. The ratio of survival (%)=(No. of survival/No. of transformation) × 100. The ratio of transformant (%)=(No. of PCR positive/No. of survival) × 100.
Table 2. Genetic transformation of M. polymorpha L. by tissue culture independent.
Figure 3: Genetic transformation of M. polymorpha L. by immersion alone, or negative-pressure infiltration. (A-J) Transformation by immersion simply; (K-T) Transformation by negative-pressure infiltration, Thalli (three weeks) transformation, Gemmae transformation, GUS staining. 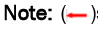 specific products in Polymerase Chain Reaction (PCR) for transformant identification;
specific products in Polymerase Chain Reaction (PCR) for transformant identification; staining sites.
staining sites.
There were no significant differences among the differential explants regarding survival ratio after selection, regardless of the method or duration of treatment employed. However, for all kinds of explants, a higher ratio of transformant was obtained by negative-pressure infiltration than by immersion alone; and increased time of treatment resulted in a higher ratio of transformant by each of these two kinds of treatment (Table 2). These transformants (T0 generation) were chimeras that were determined by β-glucuronidase staining (Table 2 and Figures 3A-3T).
Transformants (T0) that were generated from differential explants (three to four weeks old thalli or mature gemmae) show three distinct phenotypic changes. Type I: More than two new thalli developed simultaneously from the edge of the same old thallus instead of the usual dichotomous branching pattern; these new thalli could grow to enough size and be transplanted on soil for vegetative reproduction. Type II: New thalli development was based on normal dichotomous branching; however, numerous kinds of swelling structures (amorphous tissue) developed on the upper surface of the thalli; these swelling structures could evolve into curled flat structures and could not be transplanted. Type III: New thalli developed on the tip of a stalk that was originated from apical meristem; new disk-like thalli were peltate and grew in a polarity manner; these new thalli could also be transplanted. For these three kinds of transformants, gemmae development occurs on the upper surface of the gemmae cup. Expressions of the MpCCAAT-NFYB5 gene for all T0 transformants that possess phenotypic changes were higher than wild type: Type III>Type II>Type I (Figures 4A-4F).
Figure 4: Phenotypic changes and qRT-PCR analysis of MpCCAAT-NFYB5 transformants. (A-C) Phenotypic expression of type I, II, and III; (D-F) Quantification of MpCCAAT-NFYB5 for type I, II, and III transformants. Note:  various phenotypic expressions; ( T3-n) (T4-n) (G-n)No. of transformants that were transformed from three weeks old thalli, four weeks old thalli, and mature gemmae.
various phenotypic expressions; ( T3-n) (T4-n) (G-n)No. of transformants that were transformed from three weeks old thalli, four weeks old thalli, and mature gemmae.
To obtain non-chimera transformants, sub-cultures of gemmae were performed continuously. For Tn+1 generation (n=0 ~ 3), four times of sub-culture was performed repeatedly. Gemmae (80 or so), which were used to one repeat, were obtained from one gemmae cup of a Tn (n=0~3) generation individual. Gemmae for different repeats were obtained from different mother plants. For the T5 generation, four gemmae cups that were employed to obtain gemmae for each of the four sub-culture repeats originated from the same T4 mother individual. These mother plants were produced by an individual of the former generation, which developed gemmae cups and gemmae primarily among its sister individuals. For the Tn (n=2~4) generation sub-culture, a part of individuals of one repeat survived under selection conditions, revealing an amplification band in PCR and displaying local GUS detection staining. The results demonstrated that all individuals (Tn, n=1~3) were chimeras. For the T5 generation sub-culture, however, all individuals of four repeats survived under selection conditions and exhibited an amplification band in PCR (positive of PCR identification). Furthermore, the GUS staining demonstrated complete painting. These results indicated that T4 generation produced a non-chimera transformant (Figures 5A-5H).
Screening by selection buffer could reduce the amount of work required to identify transformants, such as total DNA extraction, PCR, electrophoresis, etc. All of the works reported earlier that were aimed to achieve genetic transformation using Agrobacterium-mediated transformation and particle bombardment were performed on a tissue culture basis. Hygromycin B, which was added in solid media with 10 µgmL-1 or poured on solid media (1 mL of solution with 100 µgmL-1 for each Petri dish), was frequently employed as a selective antibiotic in the genetic transformation of M. polymorpha L. Hygromycin is 20 times more toxic than kanamycin and should be utilized more effectively during selection; however, kanamycin has also proven to be effective in the selection of M. polymorpha L. Our tests about the sensitivity of M. polymorpha to kanamycin demonstrated that the Lethal Concentration (LC) is 1.0 mg·mL-1 for solution culture with 1/2 B5 medium, and 11.5 mgmL-1 for soil culture (Table 1). The increased LC of kanamycin in soil culture might be caused by the natural degradation of antibiotic molecules, absorption of soil particles, increased resistance of growing plant body, etc. Although an increased concentration of kanamycin is required for the selection of transformants in our research, effective selection could be accomplished through a simple operation without sterile tissue culture.
Pre-culture of plant materials is necessary to enable the genetic transformation of M. polymorpha L. However, collections of thalli or gemmae could be directly treated with Agrobacterium. Co-culture of explants and Agrobacterium, which is an important step of Agrobacterium-mediated transformation, could result in an excessive growth of Agrobacterium that could interfere with genetic transformation frequently as a result of sufficient nutrients from the medium. Co-culture through solution culture (1/2 B5 medium) in our work is similar to co-culture through tissue culture based on the support of artificial medium. Despite the absence of a sucrose supplement, algae could propagate rapidly and cause difficulties for subsequent procedures as a result of a non-sterile culture environment. Contrarily, co-culture through soil cultivation could be performed in our study without retardation from Agrobacterium and algae. This could be achieved from fewer nutrients supplemented by soil than artificial medium or from the sophisticated interplay between various organisms present in the soil. Normal growth in co-culture and selection steps with a lack of interference from Agrobacterium and algae facilitate subsequent operations enormously. Effective transformation of MpCCAAT-NFYB5 (Mp6g07410.1) was accomplished under these simple conditions. Over-expression of MpCCAAT-NFYB5 resulted in changes in phenotype; the phenotypic changes were consistent with those reported by Yao et al. The phenotypic changes could also be used as evidence of successful transformation. Modifications of new thalli formation in MpCCAAT-NFYB5 transformants are similar to the development of multiple leafy cotyledons in LEC1 (leafy cotyledon1) Arabidospsis thaliana [38].These results indicate that MpCCAAT-NFYB5 regulates cell division during thalli formation in M. polymorpha L.
Chimeras in plant genetic transformation occur in operations when multi-cellular explants function as receptors for exogenous genes. To avoid problems resulting from chimera, non-chimera transformants should be produced using two strategies, including single-cell transformation (i. e. protoplast or pollen) and continuous selection of offspring that are reproduced sexually from chimera transformants resulting from genetic transformation with multi-cellular explants [39,40]. For M. polymorpha L., particle bombardment or Agrobacterium-mediated method were used to determine transformations of suspension-cultured cells. Transformation of spores for M. polymorpha L. could also produce non-chimera transgenic plants. However, developing spores (sporelings), which are nascent thalli containing numerous cells in deed, were used as explants other than single cells. In this study, mature gemmae or developing thalli were adopted as explants in accordance with previous works. Gemma cup primordial are a few cells away from the apical cells [41,42]. Chimeric transformants were produced as a result of the transformation that occurred in some gemma cup primordial cells as opposed to one apical cell or entire primordia. Therefore, gemmae cultures are subsequently required for screening non-chimeric individuals. The entire process of acquiring non-chimeric transformants from the transformation of multi-cellular explants (i.e., thalli or gemmae) could consume approximately 20 weeks (four subcultures repeated continuously).
In summary, our study resulted in the development of a simplified transformation method for M. polymorpha L. based on soil culture. This method comprised immersion of explants in the solution of Agrobacterium (either with or without manual negative pressure treatment), inoculation on soil, and screening by spraying of selection buffer. In contrast to the traditional tissue-dependent protocol, this method did not involve a time-consuming tissue culture approach to obtain sterile explants. This protocol also possessed simple operations for co-culturing (explants and Agrobacterium) and selecting transformants without the requirement of sterile conditions. In addition, the acquisition of non-chimera was simple and rapid. Therefore, this non-tissue culture-dependent protocol could simplify operations of genetic transformation in M. polymorpha L. and could be beneficial for further research on the model plant.
This work was supported by the National Natural Science Foundation of China (Grant No. 31960067 and 31560081).