ISSN: 2321-6204
ISSN: 2321-6204
Dairy Chemistry Division, NDRI- Karnal (Haryana), India
Received Date: 30/03/2017; Accepted Date: 18/04/2017; Published Date: 24/04/2017
Visit for more related articles at Research & Reviews: Journal of Food and Dairy Technology
Whey protein (WP) was glycated with maltodextrin (MD) by a Maillard type reaction by dry heat treatment of lyophilized mixture of whey protein concentrate 70 and maltodextrin at 90°C for 2 hours at 80% relative humidity. Glycation was confirmed by sodium dodecyl sulfate polyacrylamide gel electrophoresis and degree of glycation using 2,4,6-Trinitrobenzene sulfonic acid method. The structural changes of WP-MD were assessed using Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy. The isoelectric point of WP was shifted towards lower pH side after glycated with MD. The conjugates had improved emulsifying properties compared to WPC and WPC-MD mixes. When assessed in the pH range 3.0-7.0, conjugates had improved solubility compared to WPC, particularly around isoelectric point of the protein. The results indicate that the WP-MD conjugates have better functional properties and can be further used as food grade wall materials in the preparation of micro/nano-emulsions in food systems.
Whey protein, Maltodextrin, Maillard reaction, Conjugates, Functional properties
Proteins are extensively used in the food industry due to their ability to add unique functional properties such as emulsifying, foaming, gelling and solubility attributes [1]. Milk proteins, particularly whey proteins, have been widely used as emulsifiers in foods [2] due to their amphipathic nature [3]. They form strong and cohesive protective film that helps prevent droplet aggregation [4]. Whey proteins consist of α-lactalbumin, β-lactoglobulin, bovine serum albumin, lactoferrins, immunoglobulins and proteose-peptones (peptides) and these contains abundant amount of essential amino acids and hence have high nutritional value. Whey protein isolate and whey protein concentrate (WPC) [5] are the two forms of whey preparations used in foods as emulsifying agents. Apart from their nutritional value, whey proteins have been studied as surfactants [6] and delivery systems of bioactive compounds such as pharmaceuticals [7] and nutraceuticals [8]. On the other hand, the functional properties of whey proteins can become poor under certain conditions due to aggregation or precipitation of protein. This effect is most pronounced at pH close to the isoelectric point of the proteins. Moreover, the increasing cost and functional demands on milk protein ingredients have created a need for speciality milk protein ingredients which possess requisite functional properties for utilization in specific food ingredients.
Maltodextrin are hydrolysis products of starch consisting of alpha-(1, 4) and alpha-(1,6) linked D-glucose polymers and/or oligomers with a dextrose equivalent less than 20. Maltodextrins (MD) are widely used in the food industry as stabilizers (texture and bulking modifiers) in food emulsions [9]. Commercial MDs of different dextrose equivalent values (DE 2-20) possess different physicochemical properties including solubility and viscosity. However, maltodextrins with the same dextrose equivalent may also possess very different physicochemical properties depending on the hydrolysis procedure and source/composition of the starch used in their preparation [10].
It has been well established that the functional properties of proteins can be further improved by covalent bonding with polysaccharides and smaller sugars [11]. Milk proteins can be modified either by physical, chemical and/or enzymatic treatments to obtain desirable functional properties [12]. Due to various safety issues, for food applications not all of these approaches are suitable for producing modified proteins. Protein– polysaccharide conjugation via a Maillard-type reaction is the best method of modification of protein which is recognized as being suitable for food applications [13].
The Maillard reaction is a spontaneous and naturally occurring reaction, in contrast to acetylation, deamidation, succinylation and other chemical methods available to improve the functional properties of proteins. The Maillard reaction can be occurred mainly in three stages; initial, intermediate and advanced. The reaction occurred in the initial stage involves the condensation of the available e-amino groups (The primary reactive amino group is lysine) of the protein with carbonyl group of the reducing Sugar, resulting in the production of amodori compounds via the formation of schiff base [14]. The intermediate stage involves the amadori compound degradation results in a wide range of products. The final stage of the reaction results in the formation of ‘’melanoidins” [15]. The dry heating method is the preferred method for protein-polysaccharide conjugation as this method is carried out at the optimum water activity and less time is required for the reaction to occur in comparison to wet heating method.
In the present investigation, whey proteins were glycated with maltodextrin by dry heat treatment method and the prepared conjugates were analyzed for SDS-PAGE, ATR-FTIR, degree of glycation, color, emulsifying properties and solubility as a function of pH.
Materials
Whey protein concentrate (WPC-70) was procured from Modern Dairy Pvt. Ltd. Karnal, Haryana. Maltodextrin (MD) was procured from Hi Media Laboratories Pvt. Ltd., Mumbai. Periodic acid, Schiff’s reagent and Trinitrobenzene sulfonic acid were obtained from Sigma Aldrich Ltd.
Preparation of Glycated Whey Protein
WPC (70) and MD were separately dissolved in distilled water at a mass ratio of 1:1 and 1:2 (WPC: MD). After hydration of 8 hours, whey protein and maltodextrin solutions were mixed and pH was adjusted to 7.0. The samples were then freeze dried to remove water, and were ground to make powder. The powder was subjected for dry heat treatment at 90°C for 2 hour at 80% RH. 80% RH was maintained using preheated desiccator containing saturated solution of KCl.
Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis (Sds-Page)
SDS-PAGE was performed according to the method of Laemmli [16] using 12% w/v resolving gel and 5% w/v stacking gel. Sample solution (6 mg protein/ml) was mixed with equal volume of sample buffer containing ß-mercaptoethanol followed by boiling for 5 min. 20 μl of prepared sample was loaded into each well and electrophoresis was carried out at constant voltage of 210 V, power of 6 W and current of 70 mA at refrigeration temperature until the indicator dye reached the gel bottom. The gel sheets were stained for both protein (0.25% Coomassie brilliant blue G-250) and carbohydrate (1% Periodic acid Schiff (PAS) solution) [17].
Color Measurement
The extent of maillard reaction that occurred in the conjugates were determined by measuring the Hunter chromaticity coordinates (L* a* b*) with Colorflex® (Hunter lab, Reston, Virginia, USA) loaded with the Universal software (version 10). The instrument was calibrated with a standard white and black tile before measurement. Dry WPC or WP-MD conjugate, was placed into a petri-dish and the L* a* b* color coordinates were measured. The light source was dual beam xenon flash lamp. In the L* a* b* color space system, L* values (lightness/darkness) ranges from 0 (black) to 100 (white), a* values (redness) ranges from -60 (green) to +60 (red), and b* values (yellowness) ranges from -60 (blue) to +60 (yellow). The color intensity (C*) was calculated using the below formula:
C*= (a2 + b2)½
Degree of Glycation
The degree of glycation was measured for unreacted amines using the 2, 4, 6-trinitrobenzene sulfonic acid (TNBS) method [18], with some modifications. A fresh working solution of TNBS was prepared prior to assays by diluting the 5% TNBS solution (Sigma- Aldrich Inc., St. Louis, MO, USA) in 0.1 M sodium bicarbonate aqueous solution to an overall TNBS concentration of 0.01% w/v. Two hundred μg/ml of WPC and each conjugate solution was prepared in 0.1 M sodium bicarbonate solution at pH 8.5. TNBS working solution was then mixed with the sample solutions at a volume ratio of solution: TNBS working solution=2:1. The mixture was incubated in water bath at 37°C for 2 h and then the reaction was terminated by adding 10% w/v SDS solution and 1 N HCl to a volume ratio of mixture: SDS: HCl=6:2:1. Absorbance of the final mixture solution was measured in triplicates at 335 nm using UV-Vis spectrophotometer with distilled water as blank.
Attenuated Total Reflectance Fourier Transform Infrared (ATR-FTIR) Spectroscopy
Spectra of samples were measured using FTIR spectrometer (IR Affinity-01, Shimadzu, Japan) with diamond crystal cell ATR and inbuilt IR-Solution software at 4 cm-1 resolution. Spectra were measured in terms of absorption. Each spectrum was averaged over 30 scans with a 4 cm-1 resolution. Spectra in the wavenumber range from 1400 to1800 cm-1, which covers the typical amide I and amide II peaks were selected for analysis.
Zeta Potential
The zeta potentials samples were measured using Malvern Nanoparticle Analyzer (Zetasizer software, version 7.03). The dispersions were prepared in deionized water at a protein concentration of 5 mg/mL adjusted to pH 3.0−7.0 using 0.1 N HCl and 0.1 N NaOH followed by filtering using a 0.45 μm syringe membrane before the analysis. Average values of three measurements were reported for each sample. Isoelectric point was determined (value equals to zero zeta potential).
Emulsifying Properties
The emulsifying properties of the conjugates were determined by the method of [19] with some modifications. Conjugate (0.7% w/v protein) was dissolved in 90 ml 0.06 M phosphate buffer, pH 7.0, containing 0.01% (w/v) sodium azide (aqueous phase) at ambient temperature under moderate magnetic stirring conditions for 1 h. The emulsions were prepared by mixing the aqueous phase with the 60 ml of oil phase (Soya bean oil) and homogenizing the mixture with an Omni GLH at 15,000 rpm for 1 min. For emulsifying activity (EA), the emulsions were centrifuged in graduated tubes at 2600 g for 5 min and the whole volume (WV) of the system and the emulsion phase volume (EPV) were measured. Emulsifying activity was expressed as:
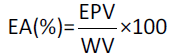
For emulsion stability (ES), the emulsions were kept at 80°C for 30 min, then in an ice bath for 15 min and finally centrifuged at 1300 g for 5 min. The emulsion stability was calculated as:

Protein Solubility
The solubility of WPC, WPC-MD mixes and whey protein-maltodextrin conjugates as a function of pH was determined according to the method of Mohanty et al. [20]. 200 mg sample was accurately weighed and dispersed (1% w/v protein) into 20 ml distilled water contained in a 250 ml glass beaker. Then pH of the dispersion was adjusted in the range of 3.0-7.0 (with 0.5 unit interval) using 0.1 N HCl or 0.1 N NaOH as required. Moderate magnetic stirring of the dispersion was carried out for 1 h. After 30 min of moderate magnetic stirring, the pH of each sample was rechecked and re-adjusted, if necessary. 14 ml of the dispersion was measured in 15 ml centrifuge tubes and centrifuged at 1000 g for 20 min. After centrifugation, the supernatant was decanted and filtered through Whatman No. 1 filter paper. The soluble and total protein content were determined using filtered supernatant and initial dispersion respectively by Bradford method [21].
Statistical Analysis
All statistical analysis was performed using MS-EXCEL-2010 package and GraphPad Prism 5.0. Results are presented as mean ± standard error (SE) of three replicates, and significance was tested by employing analysis of variance (ANOVA).
Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
Glycation of whey protein to polysaccharide was confirmed by SDS-PAGE as shown in the Figure 1. SDS-PAGE was used as characterizing tool for protein – polysaccharide conjugates as mentioned in previous studies [22-24]. Coomassie brilliant blue stain was employed to identify protein component (Figures 1 A and 1C) and periodic acid stain to identify polysaccharide components (Figure 1B). The native WPC, WPC-MD mixes (1:1 and 1:2) (without heat treatment) showed mainly three major bands at around MW of 14.0, 18.0, and 70.0 kDa that represents α-lactalbumin, β lactoglobulin and bovine serum albumin respectively. After WP glycated with maltodextrin (for both WP-MD, 1:1 and 1:2 conjugates), the native bands corresponding to α-lactalbumin and β-lactoglobulin disappeared and a smearing zone appeared at a MW of higher range than that of β-lactoglobulin and the band corresponding to bovine serum albumin was also shifted to a higher MW range. After 2 hours of conjugation, the band corresponding to α-lactalbumin was completely disappeared and this indicates that α-lactalbumin is easier to be conjugated with MD than other whey protein has previously been reported [25]. The present findings commemorates the results of Liu and Zhong [23,24]. Similarly, Gordon et al. [26] reported that conjugates of sodium caseinate with maltodextrin are confirmed by an absence of distinct bands and the formation of a diffuse band over a larger MW range although the bulk of material is in the range 25,000-37,000 g, which is within the expected spread of molecular weights taking into account the different molar masses and amounts of lysine in the individual caseins. The presence of higher MW material indicates that some aggregation or cross-linking is occurring. These results are very similar to those of Shepherd et al. [13] suggesting that conjugation and cross-linking has taken place resulting in total loss of ‘unmodified’ material and the formation of higher molecular weight material.
Figure 1: SDS-PAGE profile of whey protein concentrate, whey protein concentrate-maltodextrin mixes and whey protein-maltodextrin conjugates. The labelled lanes are: 1. WPC, 2. WPC-MD (1:1) mix, 3. WP-MD (1:1) conjugate, 4. WPC-MD (1:2) mix, 5. WP-MD (1:2) conjugate, 6. MD. A. Commasie brilliant blue stain for proteins, B. Periodic acid schiff’s stain for glycoproteins, C. Commasie brilliant blue stain with protein marker.
The findings of O’Regan and Mulvihill M [27] also shown that the conjugation leads to disappearance of characteristic native casein bands, with a distinct shift of native bands to a broad range of high molecular weight conjugated proteins. Part of protein staining material of NaCN–MD100 crude conjugate also remained at the interface of the stacking and separating gel, which indicates the presence of high molecular weight products that were too large to penetrate the separating gel. The purification of conjugates had little effect on the protein stained electrophoretic pattern. However, the large MW material that was unable to penetrate the separating gel was absent in the purified conjugate, suggesting that the material removed by purification, was primarily composed of high molecular weight Maillard reaction products.
Color
Color measurement is an indicator of the extent of advanced Maillard reaction (brown color) that occurs during conjugation of protein and polysaccharides. In the present investigation, the color coordinates (L* a* b*) of dry powders of incubated WPC, WPC-MD mixes and its conjugates were measured and their color intensity (C*) was calculated. The results of color intensity of incubated WPC, WPC-MD mixes and its conjugates are presented in Table 1 Color measurement of WP-MD conjugates generally agreed with visual observation. As indicated by Hunter chromaticity coordinates (L* a* b* values), the conjugation of WP-MD resulted in increase in a*, b* values and decrease in L* value which indicates yellow color from the Maillard reaction. Similar results was shown by Wang and Zhong [28] and O’Regan and Mulvihill [29]. As conjugates are formed via the Maillard reaction, the color of conjugates is very important for potential applications of these ingredients. O’Regan and Mulvihill [29] had shown that the conjugation of MD100 with NaCN resulted in an increase in the a* and b* values and a decrease in the L* value in comparison to the NaCN and MD100. Purification of the conjugate resulted in a decrease in both the a* and b* values and a slight decrease in the L* value in comparison to the NaCN–MD100 crude conjugate, these results indicates that purification improved the color of the conjugate product which may be attributed to the removal of some Maillard products formed during the conjugation process because of their non-adsorption to the ion exchange resin.
Table 1. Color coordinates and color intensity of whey protein concentrate, whey protein concentrate-maltodextrin mixes and whey proteinmaltodextrin conjugate.
| Samples | L* | a* | b* | C*=(a2+b2)1/2 |
|---|---|---|---|---|
| Control (WPC) | 86.77±0.033 | 0.0433±0.019 | 12.9633±0.103 | 12.9634±0.103 |
| WPC-MD (1:1) mix | 88.18±0.038 | 0.7566±0.007 | 9.8466±0.040 | 9.8757±0.039 |
| WPC –MD (1:1) conjugate | 78.14±0.049 | 4.6900±0.024 | 28.2733±0.064 | 28.6597±0.060 |
| WPC-MD (1:2) mix | 89.00±0.020 | 0.5366±0.014 | 8.7466±0.035 | 8.7631±0.035 |
| WPC-MD (1:2) conjugate | 76.15±0.020 | 5.6966±0.059 | 29.5400±0.106 | 30.0845±0.093 |
Values are Mean ± Standard Error (n=3)
Degree of Glycation
The degree of glycation was measured based on the reaction of free amines with 2, 4, 6-Trinitrobenzene sulfonic acid (TNBS). The higher absorbance value at 335 nm indicates greater amount of free amines that remained unreacted with the polysaccharide and thus a lower degree of glycation. As shown in Figure 2 conjugates had significantly (p ≤ 0.05) lower absorbance values compared to control (WPC) and WPC-MD mixes (before heat treatment) and thus higher degree of glycation. The obtained result commemorates the findings of Wang and Zhong [28]. They showed that the highest degree of glycation at pH 6.0 and higher degree of glycation leads to more heat stability due to the effect of stronger steric hindrance property of MD molecule attached to the protein molecule against protein aggregation during heating [23,24]. The maximum rate of condensation reaction is expected at weakly acidic pH [30].
Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy
Conjugation of whey proteins results in an alteration in the contents of secondary structure [23,24]. The characteristic absorption band of wavenumber 3480−3440, 3260−3270, 2960−2878 and 1350−1300 cm−1 were assigned to vibrations of −OH, −NH, − CH, and C−N stretching respectively [31]. The peaks located around 1641 cm−1 and 1532 cm−1, which belong to amide I and amide II, respectively, are characteristics of proteins [32]. It was reported that the amide I absorption band at 1639 cm−1 was represent the spectral overlap of the C=O group, coupled with in-plane −NH bending and C=N linkage [33]. The FTIR spectra of WPC, WPCMD mixes and their conjugates. The absorption band at 1520 cm−1 was attributed to a protonated amine group [34]. The main absorption bands of WPC at 1639 cm−1 (amide I) and 1529 cm−1 (amide II) shifted to 1631 cm−1 and 1539 cm−1 for WP-MD 1:2 conjugate (Figure 3A) and to 1633 cm−1 and 1533 cm−1 for WP-MD 1:1 conjugate (Figure 3B) respectively, after glycation with MD, suggesting the consumption of amino groups and the formation of Schiff bases due to the Maillard reaction. Decreases in the absorption bands at 1639 cm−1 and increases in the bands at 1529 cm−1 indicate the conversion of the primary amino groups to the secondary amine groups during the Maillard reaction.
Zeta Potential
The magnitude of zeta potential of the WPC, WPC-MD mixes and WP-MD conjugates were shown in the Figure 4. It was shown that the magnitude of the zeta potential decreased and the isoelectric point was shifted towards lower pH after whey proteins glycated with maltodextrin. The estimated isoelectric point (pH corresponding to zero zeta potential) of the WPC, WPCMD (1:1) mix, WPC-MD (1:1) conjugate, WPC-MD (1:2) mix and WPC-MD (1:2) conjugate were 5.05, 4.95, 4.05, 4.95 and 4.01 respectively. The shift of isoelectric point towards lower values after glycation was shown previously [23,24,35]. Glycation of whey proteins alters the distribution of protein surface charge, leading to decrease of isoelectric point. The decrease of isoelectric point might be due to the decrease in the content of basic lysine on protein surface after glycation with MD, which is supported by the lower magnitude of zeta potential of glycated whey proteins than WPC at pH below isoelectric point. The lowest isoelectric point of the conjugates indicates the significant changes in the protein structure that is in agreement with the highest degree of glycation [28] as discussed in the section 3.3.
Emulsifying Properties
In the present investigation emulsifying properties were studied via the formulation of 30% (v/v) oil in water emulsions. The emulsifying properties were assessed in terms of emulsifying activity and emulsion stability. It has been speculated that the chemical modification of proteins via glycation with polysaccharides may improve emulsifying properties, especially at low pH as the isoelectric point and solubility will be altered and molecular integrity maintained [36]. Shepherd et al. [13] has shown that sodium caseinate can be chemically linked with 1,4-linked polysaccharides (e.g. maltodextrin) via the Amadori re-arrangement [37] without excessive post-Amadori Maillard reactions to form novel emulsifiers with improved emulsifying activity and stability at low pH.
Emulsifying Activity
It was shown from the Figure 5A that the emulsifying activity of WP-MD conjugates were significantly (p ≤ 0.05) higher than control (WPC) and WPC-MD mixes (without heat treatment). The conjugates of WP-MD at the weight ratio of 1:2 showed the highest emulsifying activity (89.67%). This may be due to the optimum steric stabilization at the 1:2 (w/w) ratio of WP-MD. Akhtar and Dickinson [38] showed that whey proteins are highly compatible with MD at the 1:2 (w/w) ratio in the formation of conjugates with excellent emulsifying properties. Protein – polysaccharide ratio of 1:2 showed higher emulsifying activity compared to 1:1 ratio which commemorates the findings of Xie and Hettiarachchy [39]. Though the presence of some unreacted proteins in 1:1 ratio conjugate may be slightly beneficial to its emulsifying capacity, the higher polysaccharide concentrations provide better steric stabilization and thus higher emulsifying activity. Mishra et al. [40] was shown that UF WPC-Pectin complex exhibit higher emulsifying activity compared to UF WPC alone. The emulsifying ability and emulsion stability made with WPI-Dextran conjugate were also improved compared to WPI and gum arabic (an emulsifier containing naturally derived glycoproteins) [22].
Figure 5: Emulsifying properties of whey protein concentrate, whey protein concentrate-maltodextrin mixes and whey protein-maltodextrin conjugates. A. Emulsifying activity of whey protein concentrate, whey protein concentrate-maltodextrin mixes and whey protein-maltodextrin conjugates. B. Emulsion stability of whey protein concentrate, whey protein concentrate-maltodextrin mixes and whey protein-maltodextrin conjugates.
Emulsion Stability
The emulsion stability of WP-MD conjugates were also significantly (p ≤ 0.05) higher than WPC and WPC-MD mixes as in the case of emulsifying activity. As shown in the Figure 5B the conjugates of WP-MD at the weight ratio of 1:2 showed the highest emulsion stability (86.83%). In emulsion system containing both proteins and polysaccharides, proteins form an adsorbed coherent viscoelastic layer at the oil-water interface, while polysaccharide confer stability through their thickening and gelation behavior in the aqueous phase [41]. The improved stability of the conjugate stabilized emulsions in comparison to the WPC stabilized emulsions is attributed to the conjugated protein molecule forming a bulky polymeric layer than the non-conjugated protein on the droplet surface, with the MD portion extruding outwards into the continuous phase providing better steric stabilization, thus preventing droplet aggregation and coalescence [38]. The positive impact of conjugation on the retention of thymol is consistent with improved emulsifying properties of whey protein after conjugation with MD reported in the literature [38,42]. NaCN–MD conjugate stabilized o/w emulsions showed improved stability in comparison to NaCN stabilized emulsions after storage for 20 days under accelerated shelf life testing conditions [29].
Solubility
Solubility is an important functional property for proteins, which affects other functionalities. Protein solubility of WPC, WPCMD mixes and WP-MD conjugates as a function of pH from 3.0 to 7.0 was shown in the Figure 6. It was observed that conjugates had significantly (p ≤ 0.05) higher solubility over the entire pH range tested than WPC and WPC-MD mixes particularly around the isoelectric point of WPC. This might be due to the conjugation confers a protective effect against precipitation in the isoelectric region of the protein [29,43]. The obtained results corroborate the findings of Shepherd et al. [13] and is attributed to a change in the net charge of the protein and also hydration due to the attached sugar residues. Similarly O’Regan and Mulvihill [29] reported that in comparison to NaCN, NaCN–MD conjugates had improved solubility, particularly around the isoelectric pH of the protein. The improved solubility of the protein-polysaccharide conjugates are attributed to the attachment of the bulky highly hydrophilic polysaccharides to proteins [44].
Whey proteins-maltodextrin conjugates of a high molecular weight distributions were produced via non-toxic, economical and food grade naturally occurring Maillard type reaction. The increase in the molecular weight of whey proteins glycated with maltodextrin was confirmed by SDS-PAGE. Structural changes and consumption of amino group during glycation was proved by ATR-FTIR and degree of glycation respectively. Isoelectric point of whey proteins decreases towards lower pH side after glycation with MD. In comparison to whey proteins. Whey protein-maltodextrin conjugates had improved emulsifying properties and solubility over the entire pH range tested (3.0-7.0) particularly around the isoelectric point of whey proteins.
The above results indicate that the whey protein-maltodextrin conjugates have better functional properties and can be further used as a food grade wall materials in the preparation of micro/nano-emulsions in food systems.
The authors declared no conflict of interest.
We thank to Director, ICAR-National Dairy Research Institute, Karnal, Haryana (India) for providing necessary facilities, and ICAR for providing funds under CRP on Nanotechnology platform.